Absentee Ballots
Someone asked how voting has changed during COVID. Rather than rely on social media, you can get all the facts you need from the Secretary of State website. If you have not visited the site in a while, you’ll be surprised at the upgrade,
https://sos.nh.gov/elections/voters/voting-during-covid-19-state-of-emergency/
COVID-19 Vaccine Distribution Strategy
The U.S. Department of Health and Human Services (HHS) and Department of Defense (DoD) released two documents on September 16 outlining the Trump Administration’s detailed strategy to deliver safe and effective COVID-19 vaccine doses to the American people as quickly and reliably as possible.
The documents, developed by HHS in coordination with DoD and the Centers for Disease Control and Prevention (CDC), provide a strategic distribution overview along with an interim playbook for state, tribal, territorial, and local public health programs and their partners on how to plan and operationalize a vaccination response to COVID-19 within their respective jurisdictions.
The strategic overview lays out four tasks necessary for the COVID-19 vaccine program:
- Engage with state, tribal, territorial, and local partners, other stakeholders, and the public to communicate public health information around the vaccine and promote vaccine confidence and uptake.
- Distribute vaccines immediately upon granting of Emergency Use Authorization/ Biologics License Application, using a transparently developed, phased allocation methodology and CDC has made vaccine recommendations.
- Ensure safe administration of the vaccine and availability of administration supplies.
- Monitor necessary data from the vaccination program through an information technology (IT) system capable of supporting and tracking distribution, administration, and other necessary data.
On August 14, CDC executed an existing contract option with McKesson Corporation to support vaccine distribution. The company also distributed the H1N1 vaccine during the H1N1 pandemic in 2009-2010. The current contract with McKesson, awarded as part of a competitive bidding process in 2016, includes an option for the distribution of vaccines in the event of a pandemic.
– Dr. Robert Redfield, CDC Director
Detailed planning is ongoing to ensure rapid distribution as soon as the FDA authorizes or approves a COVID-19 vaccine and CDC makes recommendations for who should receive initial doses. Once these decisions are made, McKesson will work under CDC’s guidance, with logistical support from DoD, to ship COVID-19 vaccines to administration sites.
- Download the Strategy for Distributing a COVID-19 Vaccine
- Download the COVID-19 Vaccination Program Interim Playbook
- Download the COVID-19 Vaccine Distribution Process Infographic
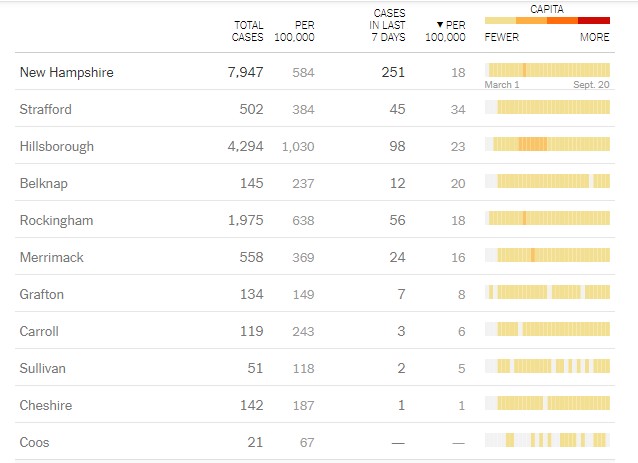
New Hampshire 2019 Novel Coronavirus (COVID-19) Summary Report
(data updated as of September 20, 2020, 9:00 AM)
| Number of Persons with COVID-19 1 | 7,947 |
|---|---|
| Recovered | 7,201 (91%) |
| Deaths Attributed to COVID-19 | 438 (6%) |
| Total Current COVID-19 Cases | 308 |
| Persons Who Have Been Hospitalized for COVID-19 | 725 (9%) |
| Current Hospitalizations | 10 |
| Total Persons Tested at Selected Laboratories, Polymerase Chain Reaction (PCR)2 | 240,401 |
| Total Persons Tested at Selected Laboratories, Antibody Laboratory Tests2 | 30,737 |
| Persons with Specimens Submitted to NH PHL | 45,489 |
| Persons with Test Pending at NH PHL3 | 768 |
| Persons Being Monitored in NH (approximate point in time) | 2,000 |
1 Includes specimens positive at any laboratory and those confirmed by CDC confirmatory testing.
2 Includes specimens tested at the NH Public Health Laboratories (PHL), LabCorp, Quest, Dartmouth-Hitchcock Medical Center, and those sent to CDC prior to NH PHL testing capacity.
3 Includes specimens received and awaiting testing at NH PHL. Does not include tests pending at commercial laboratories.









You must be logged in to post a comment.